-
Effect of Conformational Changes on a One-Electron Reduction Process: Evidence of a One-Electron PP Bond Formation in a Bis(phosphinine)
S. Choua, C. Dutan, L. Cataldo, T. Berclaz, M. Geoffroy, N. Mézailles, A. Moores, L. Ricard and P. Le Floch
Chemistry - A European Journal, 10 (16) (2004), p4080-4090


DOI:10.1002/chem.200400073 | unige:3252 | Abstract | Article HTML | Article PDF
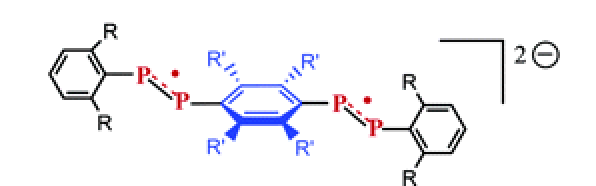
EPR spectra show that one-electron reduction of bis(3-phenyl-6,6-(trimethylsilyl)phosphinine-2-yl)dimethylsilane (1) on an alkali mirror leads to a radical anion that is localized on a single phosphinine ring, whereas the radical anion formed from the same reaction in the presence of cryptand or from an electron transfer with sodium naphthalenide is delocalized on the two phosphinine rings. Density functional theory (DFT) calculations show that in the last species the unpaired electron is mainly confined in a loose PÂ â P bond (3.479 Ã
), which results from the overlap of two phosphorus p orbitals. In contrast, as attested by X-ray spectroscopy, the P â P distance in neutral 1 is large (5.8 Ã
). As shown by crystal structure analysis, addition of a second electron leads to the formation of a classical P â P single bond (P â P 2.389 Ã
). Spectral modifications induced by the presence of cryptand or by a change in the reaction temperature are consistent with the formation of a tight ion pair that stabilizes the radical structure localized on a single phosphinine ring. It is suggested that the structure of this pair hinders internal rotation around the C â Si bonds and prevents 1 from adopting a conformation that shortens the intramolecular P â P distance. The ability of the phosphinine radical anion to reversibly form weak P â P bonds with neutral phosphinines in the absence of steric hindrance is confirmed by EPR spectra obtained for 2,6-bis(trimethylsilyl)-3-phenylphosphinine (2). Moreover, as shown by NMR spectroscopy, in this system, which contains only one phosphinine ring, further reduction leads to an intermolecular reaction with the formation of a classical P â P bond.